Formic acid is classified as a molecule with all of its atoms arranged in a single plane. A Goethe University research team, in collaboration with cooperation partners, has now experimentally proved that the atoms in formic acid jitter out of this plane on a minimal scale. The results were reported in Physical Review Letters.
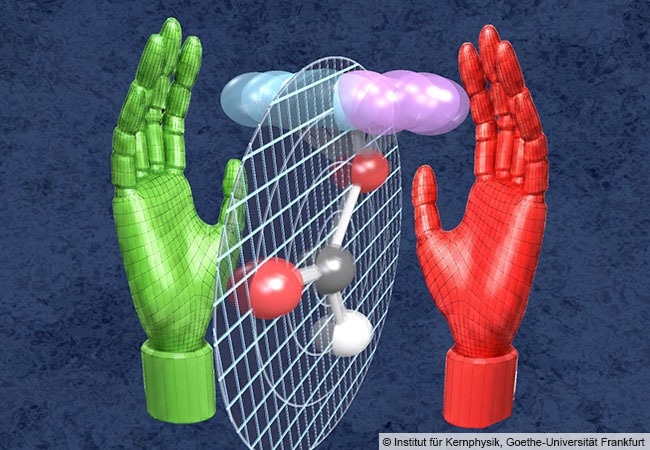 Quantum mechanical zero-point vibration, the “trembling” of the atoms, makes formic acid a chiral molecule whose two forms, like the right and left hand, cannot be superimposed. Image Credit: Institute for Nuclear Physics, Goethe University Frankfurt
Quantum mechanical zero-point vibration, the “trembling” of the atoms, makes formic acid a chiral molecule whose two forms, like the right and left hand, cannot be superimposed. Image Credit: Institute for Nuclear Physics, Goethe University Frankfurt
As a result, the molecule loses its symmetry and is often three-dimensional rather than flat. The trembling of atoms is a quantum mechanical effect, reflecting the principle that particles are never truly at rest.
Traditional chemistry textbooks paint a clean picture of molecules, with atoms occupying specific positions and stiff rods connecting them. Formic acid (methanoic acid, HCOOH) is generally thought of as a two-dimensional molecule, as flat as a sheet of paper. However, quantum physics reveals a different story. In reality, nature opposes rigidity, forcing even the most basic structures into the third dimension.
Researchers from Goethe University's Institute for Nuclear Physics, directed by Professor Reinhard Dörner, have used an X-ray beam from the PETRA III synchrotron radiation source at the DESY accelerator complex in Hamburg to identify the precise spatial structure of the “flat” formic acid molecule. They partnered with colleagues from Kassel, Marburg, and Nevada universities, as well as the Fritz Haber Institute and the Max Planck Institute for Nuclear Physics.
To do this, scientists used two reactions that occur when X-ray radiation impacts a molecule. First, the light causes the molecule to emit multiple electrons (photoelectric effect and Auger effect). As a result, the atoms become highly charged, causing the molecule to explode (Coulomb explosion). Despite the fact that they occur in femtoseconds (millionths of a billionth of a second), the scientists were able to measure them sequentially.
They employed the COLTRIMS reaction microscope, a device created at Goethe University and continually improved since then, for this purpose. They were subsequently able to determine the formic acid molecule’s initial shape using the measurement data. As a result, formic acid's two hydrogen atoms fluctuate somewhat, indicating that the molecule is not flat.
In the quantum world, atomic nuclei are not tiny spheres that remain fixed in place. They are more like vibrating clouds. Even if we cool a molecule down to absolute zero, this trembling – the so-called zero-point motion – never stops.
Reinhard Dörner, Professor, Institute for Nuclear Physics, Goethe University
The result is radical: an atomic nucleus does not have a definite position, but rather has the possibility of being discovered at a particular area. In a way, it is “a little bit everywhere”. As a result, a formic acid molecule is effectively three-dimensional at practically all times.
“Through this tiny step into the third dimension, the molecule loses its symmetry and can no longer be superimposed onto its mirror image – similar to our left and right hands. Formic acid is chiral – it has a left-handed form half the time and a right-handed form the other half,” Dörner added.
In chemistry, two such chiral forms, known as enantiomers, can have completely different effects. For example, one version of a molecule may serve as a medicine, while its mirror image may not. Normally, this handedness results from the fixed structure of a molecule.
As we were able to show using the example of formic acid, quantum trembling alone can generate two different mirror-image realities from a symmetrical molecule. This means that handedness - an important property of life - does not arise here from the molecule's static blueprint, but solely from the incessant trembling in the quantum world. More generally, our findings with formic acid show that geometry is not a static property but a dynamic event, and that a flat molecule is in reality only the average value of its atoms trembling in all directions.
Reinhard Dörner, Professor, Institute for Nuclear Physics, Goethe University
Sources:
Journal Reference:
Tsitsonis, D., et.al. (2026) Probing Instantaneous Single-Molecule Chirality in the Planar Ground State of Formic Acid. Physical Review Letters. DOI: 10.1103/bvqj-pm3n. https://journals.aps.org/prl/abstract/10.1103/bvqj-pm3n.
Goethe University