A mass spectrometer converts a gas’s partial pressure (P) into an electrical signal current (I). Its sensitivity (S) describes how strongly the instrument responds, and is defined by the relationship S = I / P.
Using this value, pressure can be computed from the signal current measurement using the formula Pa = Ia / Sa, where ‘a’ represents the gas species.
The mass spectrometer typically operates in Zone 1 (‘Z1’), where the peak width is 1 amu, as seen in Figure 2. If the current is measured at mass m, and only a single species a is present at that mass, then ImZ1 = IaZ1.
Combining the equations results in PaZ1 = ImZ1 / SaZ1, simplifying the calculation of the pressure of species ‘a’.
If two gas species possess an identical nominal mass ‘m’, then ImZ1 = IaZ1 + IbZ1. Where this is true, it is not possible to determine the individual currents or the partial pressures of ‘a’ and ‘b’.
Dual Zone Instruments
Mass spectrometers can also function in Zone H (‘ZH’), which offers significantly greater resolving power. This allows for the separation of species ‘a’ and ‘b’, which share identical nominal mass, even with an actual mass separation value as low as 0.005 amu. Each signal current is measured as IaZH and IbZH, as shown in Figure 1.
Define the ratio of signal currents as Q = IaZH / IbZH
Given that the Sensitivity Ratios in Z1 and ZH are equivalent, i.e., SaZ1 / SbZ1 = SaZH / SbZH, and as S = I / P and P is constant, it follows that,
IaZ1 / IbZ1 = Q
Therefore, IaZ1 = Q * IbZ1
Assessing the sensitivity in ZH mode is challenging. For this reason, pressures are best calculated using the signal current measured in Z1 mode, where the species sensitivity is generally known or can be readily quantified as needed.
Given that ImZ1 = IaZ1 + IbZ1
then ImZ1 = Q*IbZ1 + IbZ1
it follows that ImZ1 = IbZ1 * (1 + Q)
hence IbZ1 = ImZ1 / (1 + Q)
and IaZ1 = ImZ1 / (1 + 1/Q)
Now, the individual pressures for both species can easily be calculated,
Pa = IaZ1 / SaZ1 or Pa = (ImZ1 / (1 + 1/Q)) / SaZ1 or Pa = ImZ1 / ((1 + (IbZH / IaZH)) * SaZ1 )
Pb = IbZ1 / SbZ1 or Pb = (ImZ1 / (1 + Q)) / SbZ1 or Pb = ImZ1 / ((1 + (IaZH / IbZH)) * SbZ
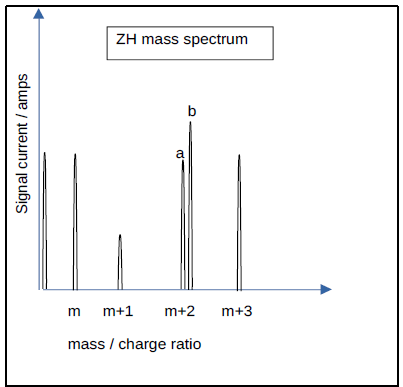
Figure 1. Image Credit: Hiden Analytical
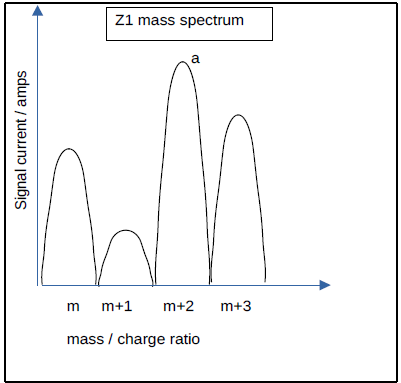
Figure 2. Image Credit: Hiden Analytical
Example: He and D2 Partial Pressure Determination Using DLS-2
This shows how Massoft displays the instantaneous computation of He and D2 partial pressures and He concentration.
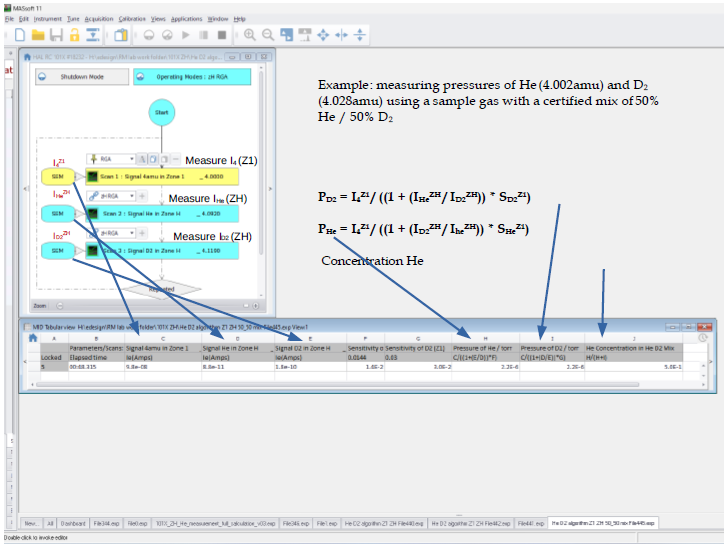
Image Credit: Hiden Analytical
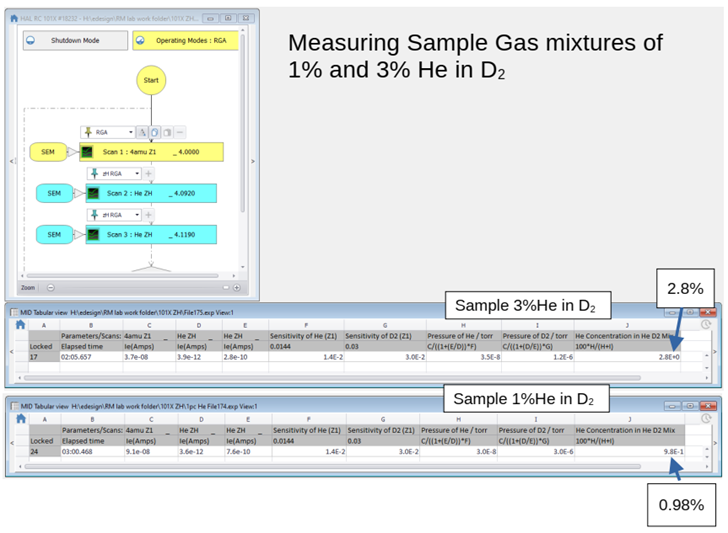
Image Credit: Hiden Analytical
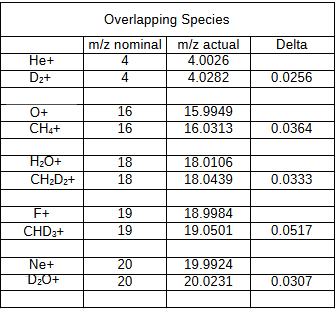
Overlapping Species Compatible with Measurement by Dual Zone Instruments
Source: Hiden Analytical

This information has been sourced, reviewed, and adapted from materials provided by Hiden Analytical.
For more information on this source, please visit Hiden Analytical.