Mar 15 2019
Chiral agents, which are contained in 7 out of 10 most common medications, are molecules occurring in right- or left-handed forms. At the time of chemical synthesis, both forms generally exist in equal parts and must be separated later, since the effect of the agent in the body is governed by its chirality.
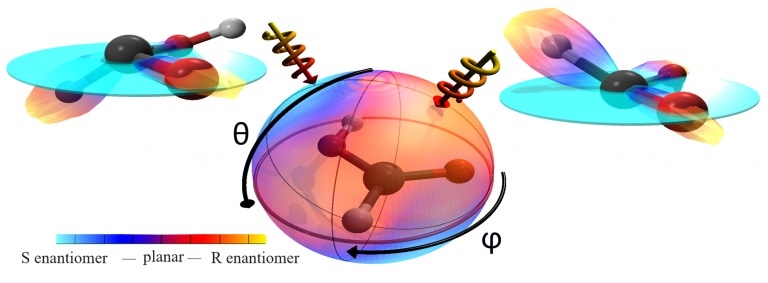 The formic acid model is in the center. The color code of the surrounding sphere shows the direct chirality of the formic acid for every direction from which the laser comes. If the laser is directed from the right side (right arrow), it results in right-handed formic acid; if from the left, in left-handed formic acid. Both chiral formic acids reflect the common structure of the molecule. (Image credit: Goethe University)
The formic acid model is in the center. The color code of the surrounding sphere shows the direct chirality of the formic acid for every direction from which the laser comes. If the laser is directed from the right side (right arrow), it results in right-handed formic acid; if from the left, in left-handed formic acid. Both chiral formic acids reflect the common structure of the molecule. (Image credit: Goethe University)
Currently, physicists from Goethe University have been successful in using laser light for creating right- or left-handed molecules.
In pharmaceutics, being able to transition a molecule from one chirality to the other using light instead of wet chemistry would be a dream.
Reinhard Dörner, Professor, Institute of Atomic Physics, Goethe University.
Kilian Fehre, Dörner’s doctoral student, has now reached a step closer to achieving this goal. He observed that the formation of the right- or left-handed version is dependent on the direction from which the initiator is hit by the laser light.
Kilian Fehre performed his experiment by using the planar formic acid molecule. He used a circularly polarized, intense laser pulse to activate it in order to transform it into a chiral form. Meanwhile, the molecule was disintegrated into its atomic components by the radiation. It was essential to annihilate the molecule for the experiment in order to be able to determine whether a mirror version or duplicate was formed.
For the analysis, Fehre employed the “reaction microscope” (COLTRIMS technique) that was built at the Institute for Atomic Physics. It enables the analysis of individual molecules in a molecular beam. The data offered by the detector following the explosive breakdown of the molecule can be used to accurately calculate the speed and direction of the fragments’ paths. This renders it feasible to reconstruct the spatial structure of the molecule.
To develop chiral molecules with the intended chirality in the future, care must be taken to ensure that the molecules are oriented similarly with respect to the circularly polarized laser pulse. This can be realized by orienting them in advance with a long-wave laser light.
The findings could also have a vital role to play in producing larger quantities of molecules with uniform chirality. Yet, the scientists presume that in cases such as these, liquids would possibly be radiated and not gases. “There is a lot of work to be done before we get that far,” considers Kilian Fehre.
The identification and manipulation of chiral molecules with the help of light is the aim of a priority program with the memorable name “ELCH” and which has been financially supported by the German Research Council since 2018. Researchers from Kassel, Marburg, Hamburg, and Frankfurt have contributed to this program. “The long-term funding and the close collaboration with the priority programme provide us with the necessary resources to learn to control chirality in a large class of molecules in the future,” concluded Markus Schöffler, one of the Frankfurt project managers of the priority program.